A chain reaction is a sequence of events where a reactive product or by-product causes additional, identical reactions to take place. In physics and chemistry, it is a self-sustaining process where one single, microscopic event releases enough energy or particles to trigger multiple subsequent events, leading to a massive, rapidly expanding cascade.
The Fascination of Chain Reactions
The concept of a chain reaction is truly fascinating. It shows how one tiny, invisible action can snowball into a massive sequence of events. Whether it’s a single domino tipping over and setting off hundreds more, a spark that spreads into a roaring fire, or the unimaginable force of a nuclear reaction, the idea that small beginnings can lead to unstoppable outcomes is both wild and inspiring.
Chain reactions remind us of the hidden power in simple causes. They teach us that even the smallest actions can create waves of change, shaping the world in ways we might never expect. From science to everyday life, this concept highlights the incredible potential locked inside the tiniest moments.
| Chain Reactions | |
|---|---|
| Scientific Field | Physics, Chemistry, Biology |
| Core Principle | Self-sustaining sequence of events |
| Key Types | Nuclear Fission, Chemical, Biological |
| First Theorized (Nuclear) | Leó Szilárd (1933) |
| Famous Example | The Atomic Bomb |
| Crucial Concept | Critical Mass |
| Controlled Use | Nuclear Power Plants, PCR Testing |
1. The Domino Effect: How It Works
The easiest way to understand a chain reaction is to imagine a meticulously lined-up row of dominoes. If you push the first domino, it falls and strikes the second, which strikes the third, and so on. You only had to provide the energy for the very first event, but the setup allowed the reaction to sustain itself until all the dominoes had fallen.
However, many scientific chain reactions are exponential. Imagine if one falling domino didn’t just hit one other domino, but instead hit two. Then those two hit four, those four hit eight, and so on. Within fractions of a second, billions of dominoes would be falling simultaneously. This exponential growth is the secret behind the terrifying power of nuclear weapons.
2. Nuclear Fission: Splitting the Atom
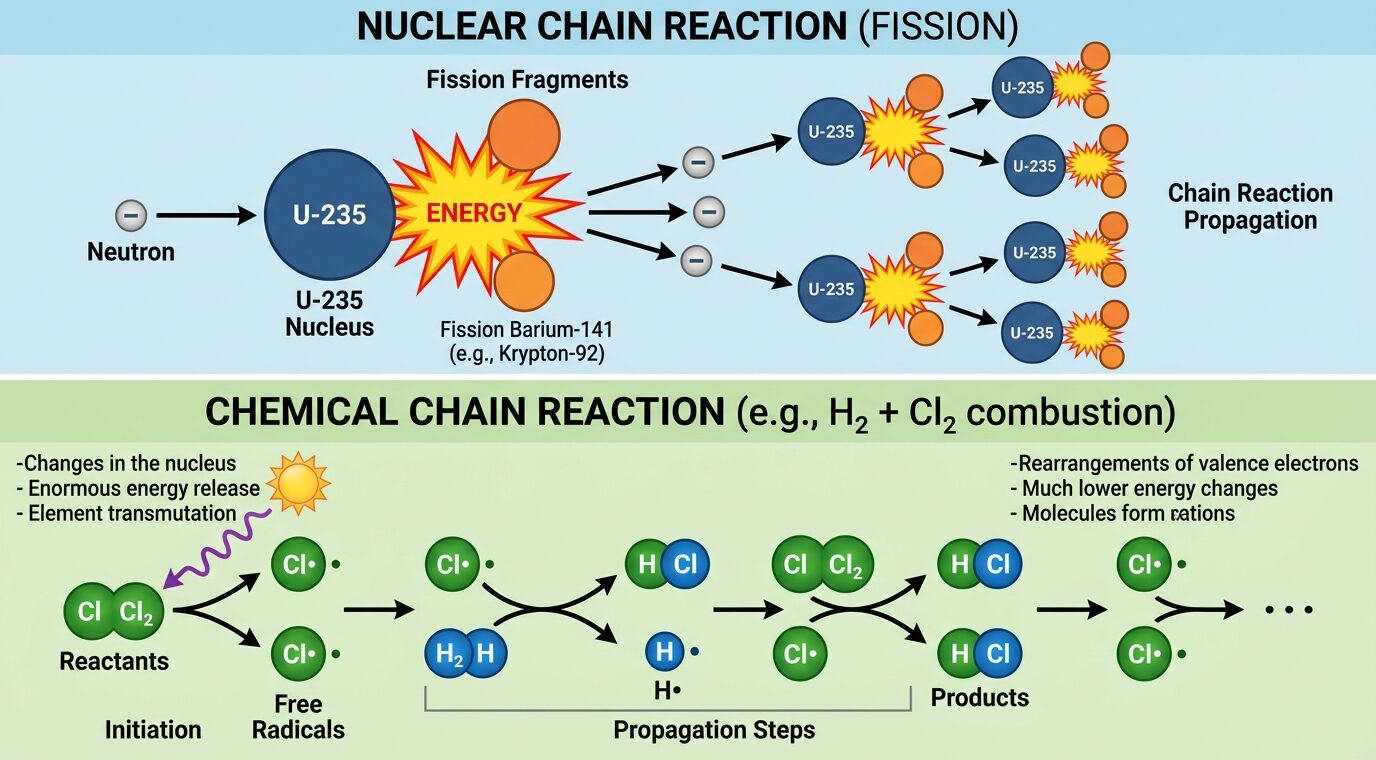
The most famous and consequential type of chain reaction occurs in nuclear physics, specifically during nuclear fission. This process involves splitting heavy, unstable atoms, most commonly Uranium-235.
- The Spark: A single subatomic particle called a neutron is fired at a Uranium-235 atom.
- The Split: The atom absorbs the neutron, becomes highly unstable, and violently splits into two smaller atoms.
- The Chain: When the atom splits, it releases a massive amount of energy. Crucially, it also spits out two or three new neutrons. Those new neutrons fly outward and strike other nearby uranium atoms, causing them to split, release energy, and fire out even more neutrons.
In an uncontrolled environment (like an atomic bomb), this exponential splitting happens in less than a millionth of a second, releasing a devastating amount of destructive energy.
3. The Concept of “Critical Mass”
Why doesn’t a piece of uranium just constantly explode on its own? It comes down to a concept called Critical Mass. For a nuclear chain reaction to sustain itself, there must be enough radioactive material packed closely together so that the escaping neutrons actually hit other atoms instead of just flying off harmlessly into empty space.
If you have less than the critical mass, the chain reaction quickly fizzles out and dies. If you have exactly the critical mass, the reaction sustains a steady, continuous burn. If you have a “supercritical mass,” the reaction accelerates out of control and explodes.
4. Controlled Reactions: Powering the World
Chain reactions aren’t just used for destruction; humanity has learned to harness them for survival. Nuclear power plants use the exact same uranium fission process as a bomb, but they heavily control the reaction. They do this by inserting “control rods” (usually made of boron or cadmium) into the uranium reactor core. These rods act like sponges, absorbing excess neutrons. By raising and lowering the rods, engineers can keep the chain reaction at a steady, safe level to boil water and generate electricity without triggering an explosion.
5. Chemical and Biological Chain Reactions
While nuclear reactions are the most famous, chain reactions happen all around us every day:
- Chemical (Fire): A simple campfire is a chemical chain reaction. The initial match provides the heat to ignite the wood. As the wood burns, it releases its own heat, which dries out and ignites the surrounding wood, keeping the fire going until the fuel runs out.
- Biological (PCR): In medicine, the Polymerase Chain Reaction (PCR) is used to rapidly copy DNA. By repeatedly heating and cooling a biological sample with specific enzymes, scientists can trigger a chain reaction that turns one single strand of DNA into billions of copies in just a few hours. This is how modern viral testing and crime scene forensics work.




