While the atomic bomb demonstrated the terrifying destructive power of the atom, humanity quickly learned how to tame this cosmic force for peaceful purposes. This process is called Nuclear Fission. Today, nuclear fission powers massive reactors that generate about 10% of the world’s electricity. It is a highly efficient, carbon-free energy source capable of powering entire cities for years on a single truckload of fuel. Understanding how we safely split atoms on Earth is crucial as the world seeks clean energy alternatives to combat climate change.
Harnessing the Power of the Atom
Nuclear energy is truly amazing because it shows how powerful science can be. The idea that we can boil water and create electricity just by breaking microscopic atoms apart feels almost unbelievable. It is a reminder that even the smallest particles in nature hold enormous energy, and when used wisely, this power can light up entire cities.
Clean energy is important for our future, and nuclear power plays a big role in that. Unlike burning coal or oil, nuclear plants can produce electricity without releasing harmful gases into the air. This makes them a strong option for fighting climate change and building a safer, healthier world for the next generations.
| Nuclear Fission Fundamentals | |
|---|---|
| Core Process | Splitting heavy atomic nuclei |
| Discovered By | Otto Hahn, Fritz Strassmann, Lise Meitner (1938) |
| Primary Fuel | Uranium-235 (U-235) |
| Major Application | Nuclear Power Plants |
| Control Mechanism | Control Rods (Boron/Cadmium) |
| Environmental Impact | Zero Greenhouse Gas Emissions |
| Primary Waste | Radioactive Spent Fuel |
1. The Science: How Do We Split an Atom?
The word “fission” means to divide or split apart. In nuclear physics, it refers to the splitting of a heavy, unstable atom into two lighter atoms. The most common fuel used for this process is an isotope of uranium known as Uranium-235 (U-235).
To initiate fission, scientists do not use physical blades or lasers; instead, they use subatomic “bullets.” A free-floating neutron is fired directly into the nucleus of a U-235 atom. When the nucleus absorbs this extra neutron, it becomes U-236, which is incredibly unstable. Within fractions of a second, the atom violently splits apart into two smaller elements (like Barium and Krypton). This split releases a massive burst of heat (kinetic energy) and gamma radiation.
2. The Chain Reaction: Sustaining the Energy
If you look at the equation above, you will notice something incredibly important: when the uranium atom splits, it not only releases energy but also spits out 2 to 3 new free neutrons. These newly released neutrons fly outward and crash into other neighboring U-235 atoms, causing them to split as well. Those atoms then release even more neutrons, creating a Chain Reaction. In an atomic bomb, this reaction is uncontrolled and exponential, leading to a massive explosion. But in a power plant, this reaction must be perfectly controlled.
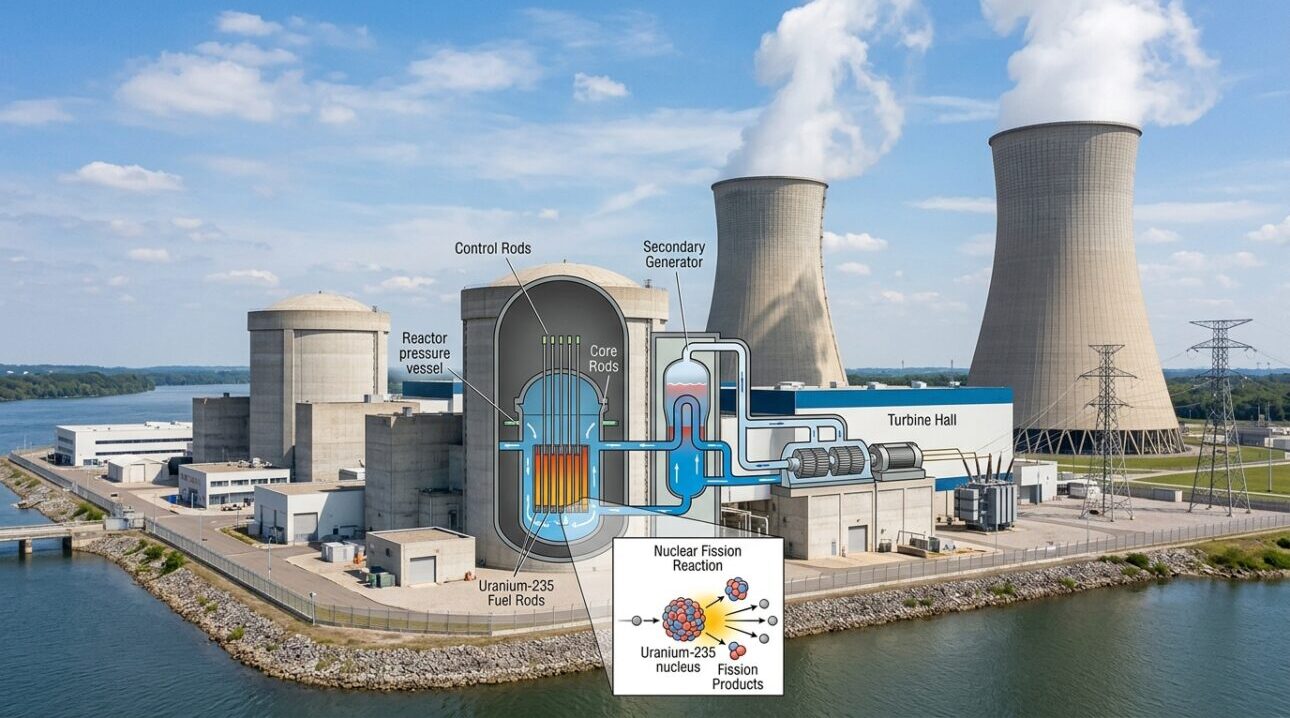
3. How a Nuclear Power Plant Works
At its core, a nuclear power plant is just a highly advanced steam engine. Instead of burning coal or gas to boil water, it uses the immense heat generated by nuclear fission. Here is how the process works:
- The Reactor Core: Ceramic pellets of Uranium-235 are stacked inside long metal tubes called fuel rods. These rods are bundled together and submerged in a giant tank of water inside the reactor core.
- Generating Heat: The fission chain reaction heats the fuel rods to extreme temperatures. The water surrounding the rods absorbs this heat.
- Creating Steam: The superheated water is pumped through pipes to a steam generator, where it boils a separate supply of water into high-pressure steam.
- Spinning the Turbine: The steam blasts through a massive turbine, spinning it at high speeds. The turbine is connected to a generator, which creates electricity that is sent out to the power grid.
4. The Crucial Role of Control Rods
To prevent the chain reaction from spinning out of control and causing a reactor meltdown (as seen in the Chernobyl disaster), engineers use Control Rods. These rods are made of materials like boron, silver, or cadmium, which naturally absorb flying neutrons without undergoing fission themselves.
By lowering the control rods into the reactor core, operators can “catch” excess neutrons and slow down the chain reaction, cooling the reactor. If the rods are pulled up, fewer neutrons are absorbed, the chain reaction speeds up, and more heat is produced. In an emergency, all control rods are instantly dropped to the bottom of the core to completely stop the fission process—a safety measure known as a “SCRAM.”
5. The Challenge: Radioactive Waste
While nuclear fission is incredibly efficient and does not produce carbon dioxide (making it excellent for fighting climate change), it does have a significant drawback: Nuclear Waste. After a few years, the uranium fuel rods are depleted. However, the leftover “fission products” inside the rods are highly radioactive and dangerous to human health. Because it can take thousands of years for this waste to naturally decay to safe levels, it must be carefully sealed in massive concrete and steel casks and eventually buried deep underground in stable geological repositories.


